Extracorporeal Shock Wave Therapy – ESWT Treatment

Extracorporeal Shock Wave Therapy
ESWT
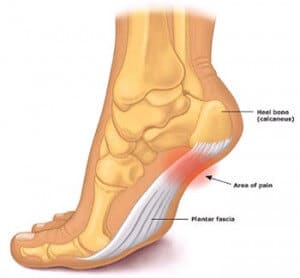
ESWT, or Extracorporeal shock wave therapy treatment is an option for patients with chronic plantar fasciitis. By delivering a focused shockwave to your heel it induces a microtrauma on the tissue that’s affected your plantar fasciitus. Your body naturally responds with a healing response that causes your blood vessel to form and increase delivery of healing nutrients to the affected area, thereby relieving symptoms of plantar fasciitis shortly after stimulating this repair process.
- 18 minute office procedure
- No risks of surgery
- Back to work same day
- Gold Standard treatment for plantar fasciitis
- No down time
PATIENT TESTIMONIALS ESWT
Read what patients are saying about Extracorporeal Shock Wave Therapy…
Plantar Fascia Pain and Extracorporeal Shock Wave Therapy
“I’ve had foot pain for over twenty years, no wonder, my job requires being on my feet eight to twelve hours a day, six days a week. Until I tried ESWT, I assumed the pain would be a part of my life forever, thankfully I was wrong.
“Working 12 hours a day in the steel mill guarantees you one thing, unbearable foot pain. The worse it got, the less I could work, until extracorporeal shock wave therapy. It has truly changed my life. In just two weeks I was pain free. It doesn’t get any better than that, unless we win the Super Bowl of course.
“I have traveled every road to try and find relief for my heel spurs-shots, taping, medicine, show inserts, losing weight and wore tennis shoes. Results-no result, until ESWT. If you don’t believe me just ask my feet.
Click Here to E-mail Dr. Christina Teimouri
Get general info on ESWT and schedule appts by calling 878-313-FEET (3338)
HOURS:
Lunchtime appointments for working people; Mon – Fri 8:00 am – 5:30 pm
Before school, after school, evening and even emergency weekend appointments to better fit your busy life.
Why Beaver Valley Foot Clinic?
- ABFAS Certified foot surgeon
- Offices available in Butler County, Beaver County and Allegheny County.
- We have treatments not offered anywhere else in Pittsburgh that save you from Surgery!
Extracorporeal Shock Wave Therapy – Pittsburgh
FAQ Frequently Asked Questions – Extracorporeal Shock Wave Therapy
How, exactly, does ESWT work?
- Device Description The Dornier Epos T M Ultra is an extracorporeal shock wave therapy (ESWT) system. The EposTM Ultra consists of a transportable cart housing the electromagnetic shock wave circuit, the hand held control unit, the CPU, a water circuit and the ultrasound subsystem. A therapy head mounted to the articulated arm, the hand control unit and the power cable are attached to the exterior of the cart. An ultrasound imaging system with a 7.5 MHz transducer is located on top of the cart. An isocentric locating arm fixed to the therapy head is used for positioning the therapy focus into the treatment area. In addition, the ultrasound is used to observe and monitor the shock wave treatment. The shock wave source of the EposTM Ultra uses electromagnetic technology to generate shock waves. Shock waves are acoustic waves that are characterized by a quick rise time of a few nanoseconds to a high maximum positive pressure (amplitude) of more than 80 Mpa (1 Mpa=10 bar). A pulse of electrical energy flowing through a disc coil at the base of the therapy head induces strong magnetic fields, which produce forces that propel the membrane producing a plane pressure wave. The shock waves travel through the water filled coupling cushion mounted to the therapy head, where they are precisely focused by an acoustic lens to the target tissue.
Why Doesn’t My Orthopedic Surgeon or Podiatrist Do This Procedure?
The physicians certified by in ESWT are truly state-of-the-art practitioners, and they are also dedicated to reducing the need for surgery. There are some surgically oriented practices that do not look to offer non-surgical treatments. You may find many physicians are not even familiar with this treatment. We encourage you to share this information with your physician and have him or her contact us for physician-oriented information.
Additionally, ESWT technology costs more than a half million dollars to offer in a practice. This investment is simply not possible within a doctor’s practice. Beaver Valley Foot Clinic owns the equipment and trains physicians in the technology and practice of ESWT. Doctors who offer this treatment are committed to avoiding unnecessary surgery and hospitalization for their patients.
Does it work? How successful is it?
- Yes, Shockwave therapy has excellent success rates when compared to other common treatments, and has saved many patients from invasive surgery. Several research papers have estimated ESWT’s success rates to be as high as 80%. In this case, success is defined as a “good” or “excellent” outcome, which means that the patient has normal function with some minor pain, or normal function without any pain at all. It often amazes patients that several extremely common treatments such as cortisone injections have practically no research supporting them, and their value is questionable at best. Dr. Teimouri in Bridgewater has been in practice for over 22 years and is very satisfied with these rates of success.
Other Treatments for Heel Pain, Pittsburgh
Orthotics and Arch Supports
What is an arch support? An arch support is foot pad or heel insert purchased OTC or by shoe size from your local podiatrist.
What is an orthotic device? An orthotic device is a custom-molded, individually designed shoe insert or ankle brace.
Orthotics to treat back pain
Back pain is very hard to treat and often multifactorial. It is influenced by many factors that we can’t control like genetics, previous injury, or work requirements.For these reasons, orthotics or heels lifts mat or may not help alleviate at least some of the symptoms and are often prescribed for back pain. A leg-length difference, which causes alterations all the way up the leg to the back is one example.
Orthotics for preventing or treating the overuse injuries of the lower body: do they make sense
Overuse injuries from walking or standing jobs, repetitive strain injuries from working out too much too soon that runners, walkers, and hikers get are a common problem.
Biomechanics are one factor, but the main problem with most repetitive strain injuries is… repetitive strain — that is, your body parts would feel the overuse and strain even if your biomechanics were fine. Our orthotics are made using a plaster cast of the foot then sent to a lab where your own personal pair of custom orthotic inserts will be created. The devices are custom made for your feet. Just as contact lenses or glasses improve vision, orthotics will help your podiatrist improve the way your foot functions.
Can ESWT be used to treat arthritis or other joint-related issues?
Extracorporeal Shock Wave Therapy (ESWT) is primarily used to treat soft tissue conditions and tendinopathies, but its application for arthritis and other joint-related issues is an area of ongoing research. Here are some key points about the potential use of ESWT for arthritis and joint-related issues:
ESWT for Arthritis
1. Mechanism of Action:
– ESWT is believed to promote healing through mechanical stimulation, increasing blood flow, and promoting the release of growth factors that can aid in tissue repair. In arthritis, these effects might help reduce pain and inflammation.
2. Types of Arthritis:
– Osteoarthritis: ESWT has been studied for osteoarthritis (OA), particularly in the knee and hip. Some studies suggest that ESWT can help reduce pain and improve function in OA patients, although more research is needed to confirm its efficacy and establish standardized protocols.
– Rheumatoid Arthritis: There is limited evidence regarding the use of ESWT for rheumatoid arthritis (RA). The inflammatory nature of RA may require different treatment approaches, and ESWT is not a standard treatment for RA.
3. Joint-Related Issues:
– Tendinopathies Around Joints: ESWT is effective for conditions like tennis elbow (lateral epicondylitis), Achilles tendinitis, and patellar tendinitis, which are common issues around joints.
– Calcific Tendinitis: ESWT is commonly used for calcific tendinitis of the shoulder, where it helps break down calcium deposits and reduce pain.
4. Evidence and Efficacy
– Research Studies: Some clinical trials and studies have shown positive outcomes for ESWT in treating knee osteoarthritis, reporting reductions in pain and improvements in joint function. However, results can vary, and more high-quality, large-scale studies are necessary to confirm these findings.
– Clinical Practice: While ESWT is not a first-line treatment for arthritis, it may be considered for patients who do not respond to conventional treatments such as physical therapy, medications, or injections.
Biomechanics, Gait Analysis & Orthotics
At Beaver Valley Foot Clinic we focus on providing the latest technology and expertise for the treatment and prevention of injuries, including:
- Radiographic and biomechanical assessment of foot and ankle disorders
- Clinical assessment of quality and causes of lower limb pain
- Prescription of corrective foot orthoses for the realignment of joints in the foot and lower limb.
- Advice and modification of diabetic insoles to offload pressure, running shoes, soccer cleats, and other sports footwear. You’ll get optimal fit based on the style of shoe you wear and an inexpensive shoe insert that rivals expensive prescription custom orthotics often costing over $600 elsewhere.